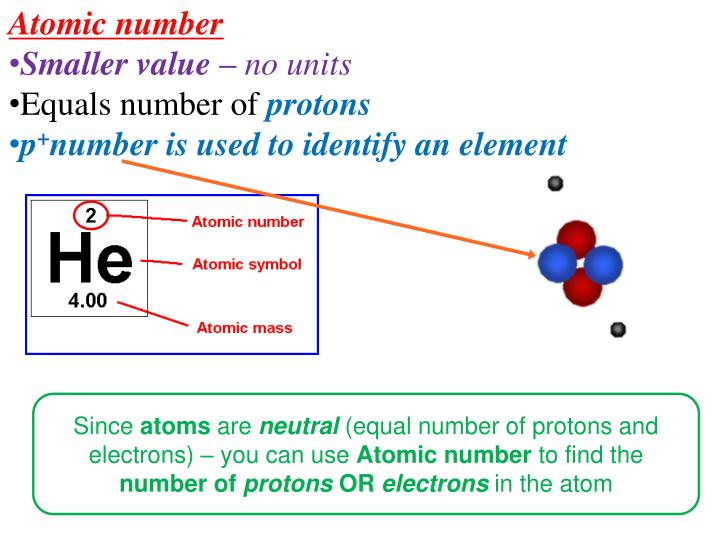
Neutron’s magnetic moment value was measured by Luis Alvarez and Felix Bloch in 1940 to be −1.93(2) μN which indicates that it has a substructure. Hughes and Burgy In 1949 reported that the angular distribution of the reflected neutrons from a ferromagnetic mirror was consistent with spin 1/2. Beta-decay is a process at which a Beta particle (electron or positron) is emitted from the atomic nucleus it can be classified into 3 types either by emission of an electron and an anti-neutrino, a positron and a neutrino, or by the capture of an electron with the emission of neutrino. The mass of the free neutron was reported to be 939.565413☐.000006meV/c 2 with a mean square radius of about 0.8×10 −15 m, the free neutron is unstable and undergoes a process called beta decay which is a type of radioactive decay at which the free neutron decays into a proton, electron, and antineutrino with a mean lifetime of (885.7☐.8 s). In 1932 James Chadwick discovered the neutron which is the neutral particle within the nucleus which was described by Rutherford. Introduction Rutherford In 1920 postulated that the nucleus consists of neutral particles and positive protons then he suggested that this neutral particle is formed from a proton and an electron combined in some way because beta radiation consists of electrons emitted from the nucleus. In fact, we will see that the Sun contains more than enough mass to destroy such huge amounts of matter and still continue shining at its present rate for billions of years.1. Destroying 4 million tons per second sounds like a lot when compared to earthly things, but bear in mind that the Sun is a very big reservoir of matter.  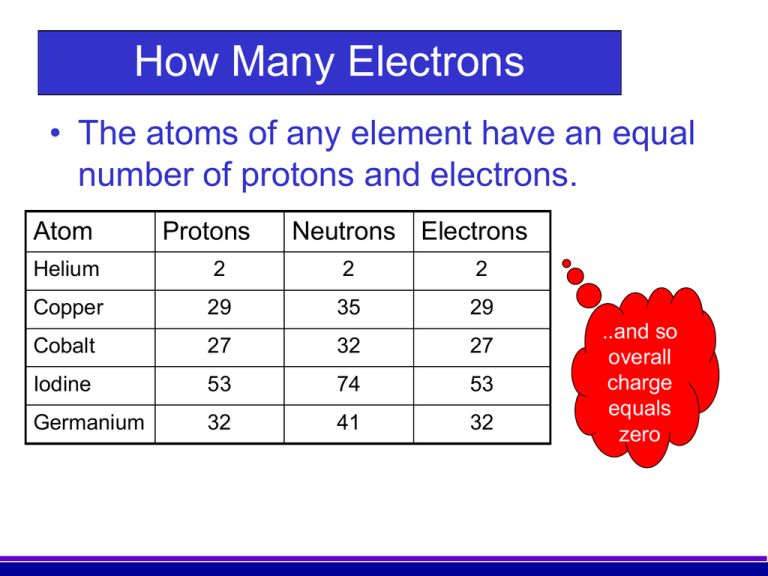
With Einstein’s \(E = mc^2\) equation, we can calculate that the amount of energy radiated by the Sun could be produced by the complete conversion of about 4 million tons of matter into energy inside the Sun each second. Scientists soon realized that the conversion of mass into energy is the source of the Sun’s heat and light. For example, the complete conversion of 1 gram of matter (about 1/28 ounce, or approximately 1 paperclip) would produce as much energy as the burning of 15,000 barrels of oil. 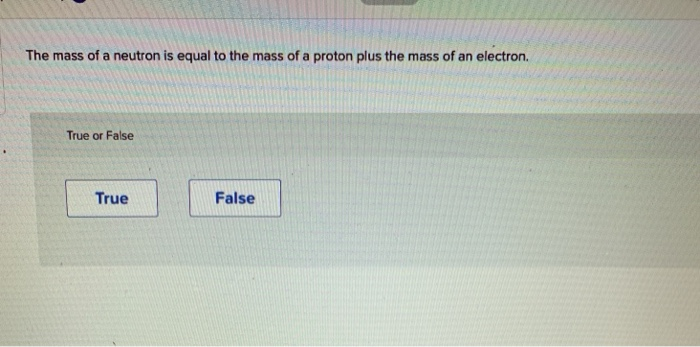
Today, as a result of developments in nuclear physics, we regularly convert mass into energy in power plants, nuclear weapons, and high-energy physics experiments in particle accelerators.īecause the speed of light squared (\(c^2\)) is a very large quantity, the conversion of even a small amount of mass results in a very large amount of energy. Einstein himself tried to discourage speculation that the large-scale conversion of atomic mass into energy would be feasible in the near future. When Einstein first derived his formula in 1905, no one had the faintest idea how to convert mass into energy in any practical way. The formulas merely tell us what the equivalent values are if we succeed in making the conversion. Notice that this formula does not tell us how to convert mass into energy, just as the formula for cents does not tell us where to exchange coins for a dollar bill. The factor of \(c^2\) is just the number that Einstein showed must be used to relate mass and energy. Note that matter does not have to travel at the speed of light (or the speed of light squared) for this conversion to occur. The conversion factor in this case turns out not to be either 12 or 100, as in our examples, but another constant quantity: the speed of light squared. Just as each conversion formula allows you to calculate the conversion of one thing into another, when we convert matter into energy, we consider how much mass the matter has.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
